Socks on Docks
-
Frequently Asked Questions
PURPOSE & BASICS
What is the purpose of the socks on docks?
The goal of this pilot is to reduce phosphorus and to a lesser degree nitrogen in Petenwell and Castle Rock lakes. Excess phosphorus is the primary driver of harmful algal blooms in freshwater lakes. This study tests whether small, distributed biochar filtration units attached to docks can help reduce phosphorus levels.
What is a sock?
A sock is a mesh sleeve filled with biochar — a carbon-rich, porous material produced by heating organic matter (wood) in a low-oxygen environment (pyrolysis). Biochar is widely studied for water filtration and is used as a soil amendment to improve soil health and sequester carbon. The socks are suspended in the water near docks, where circulating lake water passes through them. Biochar adsorbs (binds to its surface) phosphorus and certain other nutrients.
Why phosphorus? What does it have to do with algae blooms?
Phosphorus is the key limiting nutrient controlling algal growth in Petenwell and Castle Rock lakes. Small reductions in dissolved phosphorus may reduce the frequency and severity of cyanobacteria (blue-green algae) blooms, which produce toxins harmful to people, pets, and wildlife. Phosphorus enters the lake from lawn fertilizers, agricultural runoff, stormwater, and legacy phosphorus already stored in lake sediments. This pilot targets dissolved phosphorus near dock areas.
Does this filter the whole lake?
No. Dock socks are not designed to filter the entire lake. This is a distributed nutrient reduction strategy. This pilot projects will evaluate the amount of phosphorus uptake from biochar socks. Think of it as many small interventions working together, complementing larger watershed management efforts for PACRS.
Has this been tried elsewhere? Is there evidence it works?
Yes. Biochar filtration for phosphorus removal has been studied in stormwater, constructed wetlands, and agricultural drainage systems. Pilot work on dock-style or passive aquatic biochar deployment is ongoing in Wisconsin and other states’ lakes. Peer-reviewed studies confirm phosphorus sorption in controlled systems (see reference section below). This pilot contributes real-world, field-scale data to that emerging evidence base — and Wisconsin pilots like this one are helping build the case.
SAFETY & ENVIRONMENTAL IMPACT
Is biochar safe for the lake?
The biochar used in this study is lab-tested and sourced to meet environmental quality standards. It is fully contained within mesh fabric and is not loose in the water. Wood-based biochar used in aquatic filtration has been studied for nutrient removal and typically has minimal impact on lake pH when properly prepared and deployed. Nothing is left in the lake when the socks are removed.
Will this harm fish or aquatic life?
Socks are a low-impact, reversible tool designed to support — not harm — aquatic life. Algal blooms and the oxygen depletion that follows their decomposition are far more damaging to fish communities than biochar filtration. Reducing phosphorus is beneficial to fish, invertebrates, and the broader aquatic food web.
Can biochar release toxins or heavy metals?
Biochar quality depends on feedstock and production temperature. The material supplier used in this pilot is tested for contaminants prior to deployment. Certified biochar must meet strict limits for heavy metals and PAHs (polycyclic aromatic hydrocarbons) per the International Biochar Initiative (IBI) and European Biochar Certificate (EBC) standards.
Research shows that PAHs present in well-produced biochar are typically strongly bound to the carbon matrix and have very low bioavailability in water (Hale et al., 2012; Hilber et al., 2017).
Could the biochar affect pH or dissolved oxygen?
Well-prepared wood-based biochars are near-neutral to slightly alkaline in pH and are unlikely to measurably shift lake pH at the quantities deployed here. Biochar does not consume oxygen. If anything, reductions in algal biomass over time could improve dissolved oxygen conditions — decomposing algae after bloom events is a primary driver of oxygen depletion in eutrophic lakes.
What are PAHs, and should I be concerned?
PAHs (Polycyclic Aromatic Hydrocarbons) are organic compounds formed during incomplete combustion — including during biochar production. Some PAHs are carcinogens at elevated exposure. However, high-quality biochar produced at appropriate temperatures and tested to IBI or EBC standards contains PAHs well below regulatory thresholds. Research demonstrates that PAHs in certified biochar are predominantly bound within the carbon matrix and have low bioavailability in water. This is a quality-control issue, managed before deployment — not a field risk.
What about the mesh sock material itself?
The mesh fabric is selected for durability, UV resistance, and environmental compatibility for seasonal aquatic deployment. It does not leach plasticizers or harmful chemicals under normal lake conditions. The sock is fully retrieved at the end of each season — nothing is left in the lake.
HOW IT WORKS
How does biochar adsorb phosphorus?
Biochar’s highly porous internal structure creates enormous surface area — a single gram can have hundreds of square meters of internal surface. Phosphorus ions (primarily orthophosphate, PO₄³⁻) bind to this surface through electrostatic attraction and ligand exchange reactions, particularly at mineral-rich sites. The process is entirely passive: no energy input is required, and lake water circulation does the work.
What other contaminants can biochar adsorb?
Beyond phosphorus, biochar has demonstrated capacity to adsorb nitrate (to a lesser degree than P), certain heavy metals, pesticides, pharmaceuticals, and some pathogens under controlled conditions. In this pilot, phosphorus uptake is the primary target.
What happens when the dock socks fill up?
Biochar has a finite phosphorus-binding (adsorption) capacity. When saturated, effectiveness declines gradually — it does not suddenly release captured nutrients under normal freshwater conditions. A sampling of socks will be collected and analyzed to measure phosphorus capture and evaluate effectiveness at the end of the season.
How much phosphorus can one dock sock realistically capture?
Biochar phosphorus adsorption capacity varies by material and water chemistry but generally ranges from 1–30 mg phosphorus per gram of biochar in laboratory studies. Field conditions — lower phosphorus concentrations, variable contact time, competing ions — typically yield lower but still meaningful results. The pilot is designed to measure actual capture rates at a few sites. This is one of the most valuable contributions the pilot makes.
Does water temperature or seasonal variation affect performance?
Adsorption kinetics are somewhat temperature-dependent — cooler water may slow the rate at which phosphorus binds, though equilibrium capacity is less affected. Seasonal variation in lake phosphorus concentrations (often highest after spring runoff and during late-summer bloom periods) will also influence sock performance. The June–September deployment window captures the highest-risk bloom season, which is the right scientific focus.
DEPLOYMENT & LOGISTICS
How do I hang the sock?
PACRS will provide nylon rope to hang the sock vertically or horizontally from your dock or boat lift. The WI DNR requires that the sock DOES NOT touch the bottom of the lake. Placing it under the dock or along the inside of the supports protects it from incidental boat or fishing damage. Any orientation that keeps it fully submerged in the water column is effective.
What if the sock is damaged by fishing hooks, boats, or wildlife?
The sock fabric is manufactured to resist puncture, but if damage occurs, any biochar released will not harm the lake or aquatic life.
Can we swim next to the biochar sock?
Yes. Biochar and the nutrients it captures remain bound within the sock. There is no risk to swimmers from the biochar itself. Our hope is that reduced algal blooms over time will make swimming safer and more enjoyable — cyanobacteria bloom toxins (microcystins) are a genuine hazard to swimmers and pets, and reducing phosphorus is how we address that.
What do I do with the dock sock when I remove it for winter?
Handle with care — a saturated sock will be significantly heavier than when dry. Allow it to dry in a ventilated area away from storm drains or waterbodies, as drainage from the sock could carry concentrated phosphorus.
Store in a protected, dry location until spring. Lab testing will determine whether socks can be redeployed next season or should be retired. Spent biochar that passes contaminant testing can be land-applied as a slow-release soil amendment — or disposed of in household garbage if testing indicates retirement is appropriate.
How long will the pilot run?
Socks will be deployed May through September in 2026 and 2027. Other Wisconsin lakes are running parallel pilots including the Town of Rome Tri-Lakes which drains into Petenwell, enabling comparison across different lake types and watershed conditions.
Is this a permanent change to the lake?
No. This is a pilot study using a reversible, adaptive management strategy. Socks can be removed at any time if results do not support continued use. Nothing is permanently added to the lake. All materials — including the socks themselves — are fully retrieved at season’s end.
CONTACT & RESOURCES
Direct questions to PACRS
Visit the Socks on Docks page on the PACRS website: https://pacrs.org/socksondocks
Phosphorus & Nutrient Removal
Citation
Key Finding
Yao et al. (2013). Engineered biochar reclaiming phosphate from aqueous solutions. Water Research, 47, 3959-3968.
P adsorption capacity and stability; nutrient-loaded biochar as slow-release fertilizer.
Mohanty et al. (2018). Biochar for nutrient removal from stormwater: A review. Chemosphere, 200, 223-233.
Nutrient removal effectiveness across water systems.
Ulrich et al. (2015). Removal of nutrients and metals from stormwater using biochar. Journal of Environmental Quality, 44, 646-653.
Field-scale filtration performance; minimal pH disruption.
Liu et al. (2019). Biochar as a sustainable water treatment media: A review. Science of the Total Environment, 659, 588-600.
Comprehensive review of water purification applications.
Akhil et al. (2021). Production, characterization, activation and environmental applications of engineered biochar. Environmental Chemistry Letters, 19, 2261-2297.
Engineered/activated biochars with enhanced P-sorption capacity.
Contaminants, PAHs & Safety
Citation
Key Finding
Hale et al. (2012). Sorption of PAHs to biochar. Environmental Science & Technology, 46, 2830-2838.
PAHs in biochar strongly bound; low bioavailability in water.
Hilber et al. (2017). PAHs and potentially toxic compounds in biochar. Environmental Science & Technology, 51, 12577-12586.
Contaminant thresholds and role of production temperature.
IBI (2015). Standardized product definition and testing guidelines for biochar.
Global standard for contaminant limits and testing protocols.
European Biochar Certificate (EBC). Current edition. EBC guidelines for biochar quality.
Contaminant thresholds and quality assurance standards.
Aging, Stability & Long-Term Behavior
Citation
Key Finding
Cheng et al. (2006). Natural oxidation of black carbon in soils. Geochimica et Cosmochimica Acta, 70, 614-626.
Surface oxidation over time often increases sorption capacity.
Spokas et al. (2012). Biochar: A synthesis of agronomic impact beyond carbon sequestration. Journal of Environmental Quality, 41, 973-989.
Environmental behavior, soil interactions, and risks overview.
Crombie et al. (2013). Effect of pyrolysis conditions on biochar stability. GCB Bioenergy, 5, 122-131.
H:Corg ratio as proxy for carbon permanence and biochar stability.
Aquatic & Watershed-Scale Applications
Citation
Key Finding
Beesley et al. (2011). Biochars' potential role in remediation and restoration of contaminated soils. Environmental Pollution, 159, 3269-3282.
Multi-contaminant sorption and ecosystem restoration potential.
Beck et al. (2011). Biochar reduces bioavailability of trace elements in stormwater. Journal of Soils and Sediments, 11, 1073-1079.
Trace metal adsorption in stormwater; relevant to multi-contaminant filtration.
Rajapaksha et al. (2016). Engineered/designer biochar for contaminant removal. Chemosphere, 148, 276-291.
Targeted biochar design for specific contaminants including phosphorus.
Carbon Sequestration & Climate Benefits
Citation
Key Finding
Lehmann et al. (2021). Biochar in climate change mitigation. Nature Geoscience, 14, 883-892.
Foundational review of biochar as CDR strategy; permanence and co-benefits.
Woolf et al. (2010). Sustainable biochar to mitigate global climate change. Nature Communications, 1, 56.
Estimates global CDR potential of biochar at scale.
WHAT HAPPENS TO "SPENT" BIOCHAR FROM DOCK SOCKS?
"Spent" biochar simply means the material has reached or approached its phosphorus adsorption capacity. This is planned for and managed.
Key points:
1. Phosphorus is not suddenly released. Adsorption capacity declines gradually. Significant release is unlikely under normal, well-oxygenated lake water conditions.
2. Spent socks are collected and analyzed to measure phosphorus capture and evaluate performance.
3. After removal, spent biochar can be managed in several science-supported ways:
Option A: Beneficial Land Application (Preferred)
If testing confirms safety, spent biochar can be land-applied as a soil amendment. Captured phosphorus becomes a slow-release nutrient, closing the nutrient loop. Phosphorus removed from the water column returns to soil, where it benefits plant growth rather than algal growth. Supported by Yao et al. (2013) and consistent with circular nutrient management principles.
Option B: Compost Integration
Spent biochar can be blended into compost systems to enhance nutrient retention, reduce leaching, and improve compost quality.
Option C: Controlled Disposal
If monitoring detects elevated contaminants (unlikely with certified material), disposal follows standard municipal waste protocols. Household garbage disposal is appropriate for spent biochar that passes contaminant thresholds.
Key message: Spent biochar is not hazardous waste. It is a nutrient-loaded soil amendment with documented agricultural value.
“When the socks reach capacity, they’re removed and tested. The captured phosphorus can often be recycled back into soils as a slow-release nutrient — closing the loop and turning a lake problem into a soil health and long-term carbon storage tool. Nothing is left in the lake."
This FAQ was adapted for PACRS for Petenwell and Castle Rock lakes with permission granted from a FAQ written by Kristin Foehringer for a Lake Wissota dock socks project.
Our water group, Petenwell and Castle Rock Stewards (PACRS) is excited to tell you about the use of “Socks on Docks” to assist in removing phosphorus and nitrogen (nutrients that cause algae blooms) from our lakes. This letter is meant to be informative about these biochar socks and how PACRS are going to study their effectiveness in taking nutrients out of Petenwell and Castle Rock lakes.
Socks on Docks are sacks filled with biochar, which is wood that has been burnt, somewhat like charcoal. When placed into the water it absorbs the nutrients (nitrogen and phosphorus) which is the fuel for algae growth in our lakes. These bags hang off piers, without touching the lake bottom, for the summer season and are removed in the fall. They last for 2-3 years. At that time the biochar sock contents can be put into the ground to release the nutrients and carbon back into the earth. The bags cost $25.00 each.
PACRS is cautiously optimistic about how these biochar socks could supplement other conservation actions that we support from farmers and homeowners around the lakes. We are going to support testing on their effectiveness in the Southview Shores HOA this year with guidance from the DNR. Some members in PACRS are ready to purchase them for their docks ahead of the test results. 14 Mile Water Association (includes the Tri-Lakes) members will be implementing this on their lakes this year as well.
Presently, we cannot estimate the impact these socks would have on large bodies of water like Petenwell and Castle Rock Lakes. PACRS just wanted to inform you that we are gathering information and support testing this year. If you or your HOA would like to purchase Biochar socks to try at your dock, click this link.
We will place a larger order with others to reduce impact of freight on the costs to our area. We feel confident that Socks on Docks can help to pull nutrients out of our lakes and possibly decrease algae blooms but currently can only provide antidotal evidence.
Please consider trying them on your dock. You may also consider becoming a member of PACRS. https://pacrs.org/get-involved
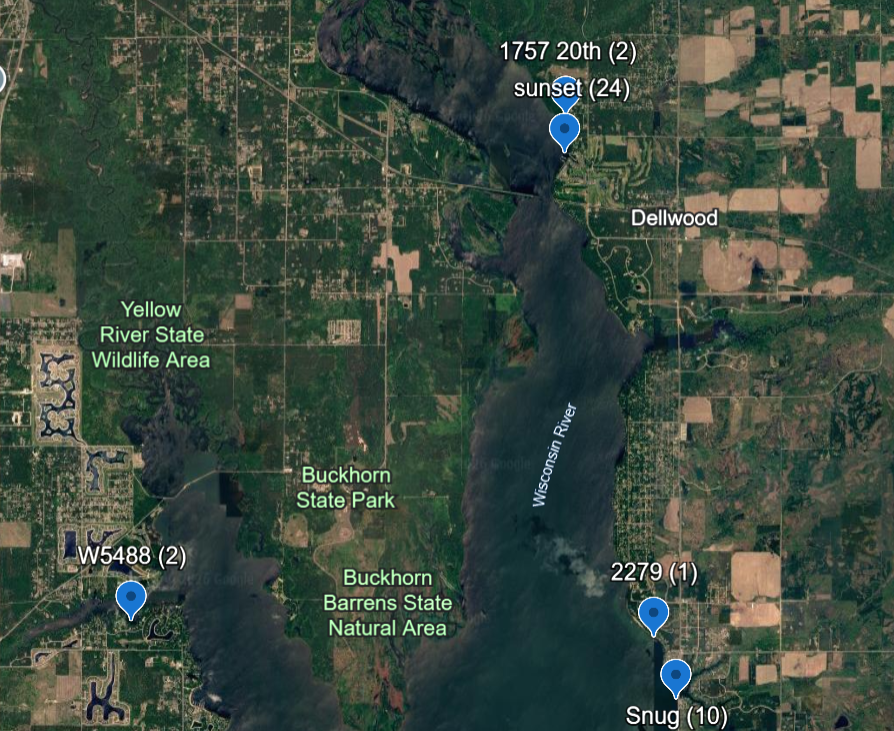
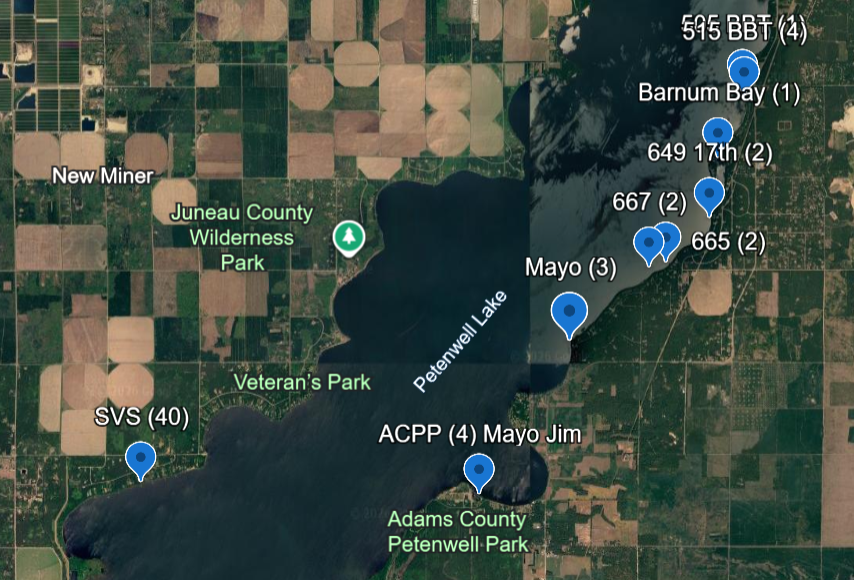
Click on link for current locations with quantities.
https://earth.google.com/earth/d/1ce0NSdkTb-n5JVdsWp0LEUrYr3cUGlhx?usp=sharing
-
Deploying Socks on Docks
Attach the sock to the dock (with 4’ rope provided) so that it is fully submerged — at least 6 inches below the water surface and not touching the lake bottom.
Choose a location with good water circulation, not a stagnant dead zone. A position perpendicular to the prevailing wave direction is ideal. Avoid areas with heavy aquatic vegetation that could clog the sock mesh.
Vertical Deployment (Hanging) Horizontal deployment is acceptable too!
Attach a rope to the sealed top end of the sock.
Tie off the rope to a dock cleat, piling or underside of dock so the sock hangs vertically below the dock, with the top end closest to the surface.
The sock bottom should be at least 6 inches above the lake bottom to allow water flow.
End-of-Season Removal & Redeployment
Dock owners remove their own regular socks at season end.
Socks should be stored in a dry location until next season or can be used as a soil amendment. Do not dispose of socks in the lake, in storm drains, or in the garbage. They can be spread on the soil that will not drain directly into a body of water.
What do I do with the dock sock when I remove it for winter?
Handle with care — a saturated sock will be significantly heavier than when dry. Allow it to dry in a ventilated area away from storm drains or waterbodies, as drainage from the sock could carry concentrated phosphorus.
Store in a protected, dry location until spring. Spent biochar land-applied as a slow-release soil amendment — or disposed of in household garbage but not in a body of water.
Is this approach recognized or approved by water quality regulators?
The WI DNR is aware of and supportive of socks on docks pilot work on Petenwell, Castle Rock and other lakes in Wisconsin. Biochar is a well-recognized soil amendment and water treatment material. This pilot does not require permits for deployment given its passive, non-chemical, and fully retrievable design. DNR coordination has informed placement guidelines (e.g., the no-bottom-contact rule). Results from this and similar pilots will inform future regulatory guidance and potential scale-up.
Images are approximate locations of socks on docks
Petenwell above and Castle Rock below